If you work with optical coatings, specialty alloys, or luminescent materials, you've probably come across lanthanum fluoride. It's one of those rare earth compounds that shows up in surprising places—from the electrodes in cinema projectors to the anti-reflective coatings on infrared lenses.
What Is Lanthanum Fluoride?
Lanthanum fluoride (LaF3) is a white to off-white crystalline powder. It won't dissolve in water, but it will dissolve in strong acids—a useful trait to know if you're working with it in the lab.
Quick identifiers:
- CAS: 13709-38-1
- EINECS: 237-252-8
- Formula: LaF₃
- Appearance: White to off-white powder
You can make it a couple of ways. The most common route is reacting lanthanum chloride or nitrate with hydrofluoric acid. Another method passes hydrogen fluoride gas over heated lanthanum oxide. Both yield anhydrous LaF3, though the choice depends on your purity requirements and available equipment.
Why Fluoride? A Quick Comparison
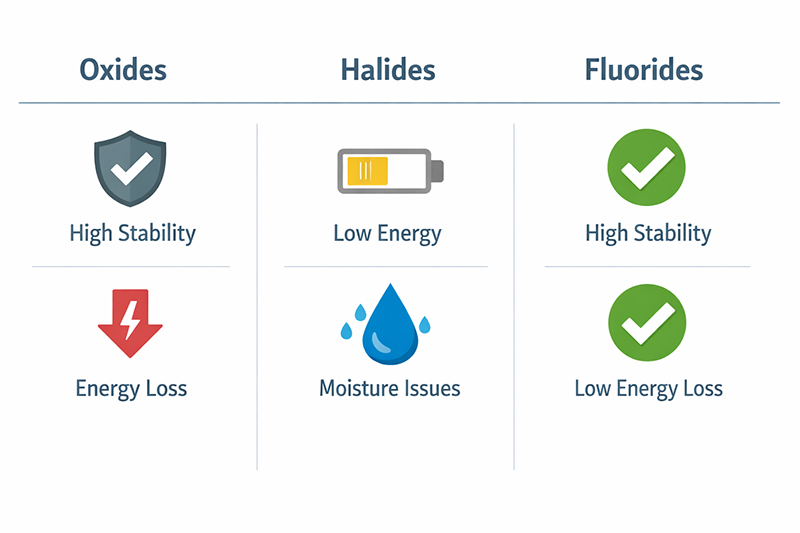
Researchers often choose between oxides, halides, and fluorides for optical and luminescent applications. Each has trade-offs.
Oxides (like Y2O3, La2O3) are stable and easy to work with. But they have high "phonon energy"—a property that causes energy loss in optical applications. Basically, they waste some of the light you're trying to capture or emit.
Halides have low phonon energy, which is good for optics. Unfortunately, most of them absorb moisture from the air and dissolve in water. That makes them impractical for real-world devices.
Fluorides sit in the sweet spot. Lower phonon energy than oxides, better stability than halides. That's why materials like LaF₃, YF₃, and NaYF₄ show up so often in upconversion and downconversion research. NaYF₄ gets most of the attention for efficiency, but LaF₃ remains popular for applications where specific optical properties or thermal behavior matter more.
At Stanford Materials Corporation (SMC), we supply LaF3 in whatever form makes sense for your process—powder if you're doing synthesis, sputtering targets if you're coating optics, evaporation materials if you're running thermal deposition.
How Lanthanum Fluoride Is Made
The preparation method depends on what you're trying to end up with.
For standard powder:
- Precipitation: Lanthanum salt solution + hydrofluoric acid → LaF₃ precipitates out
- Gas reaction: La₂O₃ + HF gas (heated) → anhydrous LaF₃
For nanoparticles or controlled morphologies:
Researchers have developed quite a few techniques over the years:
- Hydrothermal synthesis (good for crystal growth control)
- Microemulsion (for uniform particle size)
- Sol-gel processing
- Microwave or ultrasonic methods
- Precursor pyrolysis
- Electrospinning
Each method gives you different particle shapes and sizes. Which one you pick depends on your application—whether you're coating a lens, doping a crystal, or mixing into a composite material.
Where Lanthanum Fluoride Gets Used
1. Optics and Photonics
This is probably LaF3's biggest playground.
Fluoride glass fibers. You'll find LaF3 in heavy metal fluoride glasses like ZBLAN. These fibers transmit light from UV all the way to mid-infrared—much broader range than standard silica fibers. That matters for infrared spectroscopy, laser surgery, and remote sensing.
Anti-reflective coatings. LaF3 thin films reduce reflection on infrared optics. For this application, you need consistent evaporation materials—which is where SMC's LaF3 evaporation materials come in. The purity and density of the starting material directly affect film quality.
Scintillators. Radiation detectors for medical imaging (PET scanners) and nuclear security often use LaF₃-based materials.
Lasers. Doped LaF3 crystals work as gain media for certain UV and mid-infrared laser systems.
2. Metals and Alloys
Making lanthanum metal. Molten salt electrolysis with LaF3 as feedstock produces metallic lanthanum for specialty alloys.
Electrolyte baths. When producing mischmetal or rare earth alloys (lanthanum-cerium, praseodymium-neodymium), LaF3 provides the fluoride ions needed for stable electrolysis.
Calcium reduction. For metals like samarium and europium, fluorides actually work better than oxides—higher yield, cleaner separation.
3. Chemical Analysis
Ion-selective electrodes. Remember those fluoride tests for drinking water? The electrode inside uses a LaF3 single crystal membrane. Fluoride ions passing through generate a voltage proportional to concentration. Simple, reliable, and LaF₃ is the only material that does it well.
4. Lighting
Arc lamp electrodes. Add a little LaF3 to carbon electrodes, and high-intensity arc lamps run more stable. Cinema projectors, searchlights, solar simulators—they all use this trick.
Product Forms: What You Can Actually Buy
Different processes need different physical forms. Here's what Stanford Materials Corporation offers:
| Form |
What It's For |
Typical Specs |
| LaF3 Powder |
Chemical synthesis, phosphor prep, metallurgical feed |
99.9-99.99%, controlled particle size |
| LaF3 Sputtering Targets |
Thin film deposition (optical coatings, electronic devices) |
Dense, various sizes, can be bonded |
| LaF3 Evaporation Materials |
Thermal or e-beam evaporation for optics |
Granules, pellets, tablets |
If you need something specific—different dimensions, custom purity, bonded targets—just ask. We do custom fabrication regularly.
Other Rare Earth Fluorides We Carry
Lanthanum is just one of the rare earths. We supply fluorides across the whole series:
Need specs or pricing? Reach out to our sales team.
Quick Q&A
Q: Powder vs. sputtering target—what's the difference?
A: Powder is raw material—use it for reactions or as feed for thermal evaporation. Sputtering targets are dense pieces designed for PVD systems. You mount them in the chamber, and ion bombardment knocks atoms loose to deposit as thin films.
Q: Why LaF₃ for optical coatings?
A: Three reasons: it's transparent from UV to IR, has low refractive index, and holds up well environmentally. That combination is hard to beat.
Q: Can you do custom sizes for sputtering targets?
A: Yes. Custom dimensions, bonding to backing plates, special packaging—we handle it regularly.
Q: What purity do you offer for evaporation materials?
A: Typically 99.9% to 99.99%. If you need higher, let us know.
Q: Is LaF₃ dangerous?
A: Handle it with normal lab precautions. Don't ingest or inhale. Check the SDS for specifics.
Bottom Line
Lanthanum fluoride isn't the flashiest material out there. But it does things few other compounds can—stable fluoride chemistry, good optical properties, and reliable performance across a range of applications.
At Stanford Materials Corporation (SMC), we've been supplying LaF3 for years. Powder for synthesis, sputtering targets for thin films, evaporation materials for optics—we can likely match whatever form your process needs.
Contact our technical team if you want to discuss your application or request a quote.

















